 This type of error is called lack-of-fit error and cannot be reduced by increasing the number of standard replicates. The second scenario is that the curve model may not approximate the true curve very well. This type of error is designated as pure error and can be reduced by increasing the number of replicates of each standard point. The first scenario is the presence of random variation in the data set. In general, there are two scenarios where a curve will not fit the data points perfectly. The output of the curve fit optimization also includes a report to indicate which curves in a data set have been successfully optimized, could not be optimized, or require user input to further refine data points (Figure 4). The standard performance criteria used to refine individual standard curves include identifying outliers that demonstrate poor standard curve recovery, poor intra-assay %CV, and saturation or hook effect. 
This decreases total analysis time and increases the consistency in qualifying raw data. Multiple data sets can be simultaneously optimized without cycling through several screens to identify outlier data points. Software can optimize curve fit by refining the standard curve fit, automatically removing standards from analysis based on recovery values, statistical relevance, and a conservative coefficient of variation.īio-Plex Manager software includes a curve fit optimizer, a single click feature that allows users to optimize curves without manually mining the raw data. Therefore proper management of the standard curve is the key to consistent reporting of sample measurement. The quality of the standard curve has a direct impact on the quality of the extracted concentration values. A regression analysis is performed to derive an equation that is used to predict the concentrations of unknown samples. The Bio-Plex assays use serially diluted standards to generate a calibration or standard curve. Intra-assay (within run) %CV assesses variation among the replicates within the assay. Assay precision evaluates both intra-assay %CV and inter-assay %CV. Working assay range is defined as the range between the lower limit of quantitation (LLOQ) and the upper limit of quantitation (ULOQ) in which an assay is both precise and accurate. The output is derived from the assigned (expected) concentration of each standard and the actual (observed) concentration using the formula: 
The measurement can be performed automatically by software. 
This is divided by the expected concentration and multiplied by 100 to give a recovery percentage. For each analyte standard, an observed concentration is back-calculated by plotting the fluorescence intensity of the standard on the standard curve. Bio-Plex Manager includes a mechanism for assessing the fit of a standard curve to its individual standard points. Standard curve recovery is a practical parameter commonly used to determine the overall accuracy of an assay (Wadhwa and Thorpe, 1998). Critical parameters for an optimized standard curve. In the context of multiplex immunoassays, a good curve is defined by three main parameters: standard curve recovery, intra-assay CV, and working assay range (Figure 2).įig 2. 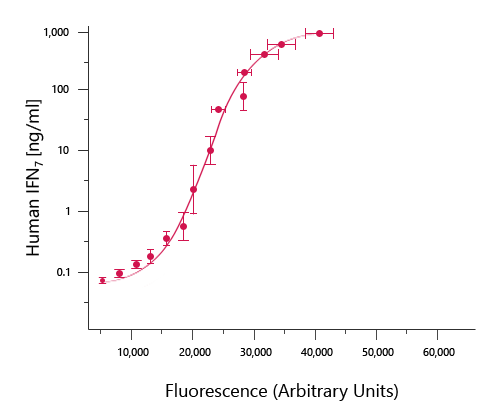
Collection settings or protocols are set up in the software to ensure that the data collected are optimized for the specified assays. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
